Developing A Treatment Plan
In cancer care, different types of doctors often work together to create a patients overall treatment plan that combines different types of treatments. This is called a multidisciplinary team. This team is usually led by a urologist, a doctor who specializes in the genitourinary tract, or a urologic oncologist, a doctor who specializes in treating cancers of the genitourinary tract. Cancer care teams include a variety of other health care professionals, such as physician assistants, nurse practitioners, oncology nurses, social workers, pharmacists, counselors, dietitians, and others.
Treatment options and recommendations depend on several factors, including:
-
The type, stage, and grade of bladder cancer
-
Possible side effects
-
The patients preferences and overall health
Your care plan also includes treatment for symptoms and side effects, an important part of cancer care.
The first treatment a person is given for advanced urothelial cancer is called first-line therapy. If that treatment stops working, then a person receives second-line therapy. In some situations, third-line therapy may also be available.
Adjuvant systemic therapy is treatment that is given after radical surgery has been completed. In bladder cancer, adjuvant therapy is usually cisplatin-based chemotherapy or treatment in a clinical trial. Neoadjuvant therapy is treatment that is given before surgery, such as cisplatin-based chemotherapy.
Treatments by type and stage of bladder cancer:
Section : Clinical Questions Related To The Intravesical Bcg Immunotherapy
Question 13: What are the contraindications of intravesical BCG immunotherapy?
Recommendation: Intravesical BCG immunotherapy is contraindicated to patients with visible haematuria, symptomatic urinary tract infection, recent history of traumatic catheterization, active tuberculosis, severe immunosuppression , allergy to BCG, and operations within two weeks of TURBT. .
Evidence summary: We referred to the recommendations from the EAU guideline , NCCN guideline , and Guidelines for Diagnosis and Treatment of Urology and Andrology in China .
Question 14: Is intravesical BCG immunotherapy prior to intravesical chemotherapy in patients with NMIBC?
Recommendations: For patients with high-risk tumors, intravesical BCG immunotherapy is recommended. . For patients with intermediate-risk tumors, intravesical chemotherapy or intravesical BCG immunotherapy is recommended.
Implementation consideration: Treatment schemes for intravesical BCG immunotherapy: starting intravesical BCG instillation within 24 weeks after TURBT The patients should first be given BCG induction instillation for 68 weeks , followed by BCG maintenance instillation for 13 years , and then repeat the treatment every 6 months .
Question 15: Is a standard dose of BCG immunotherapy superior to a low dose of BCG immunotherapy for the patients with intermediate-risk and high-risk NMIBC?
Question 18: What is the treatment option after the intravesical BCG immunotherapy failed?
Table 2 Management of BCG side effects
Monotherapy Combination Therapy And Multi
Combination therapies based on immunomodulators such as checkpoint inhibitors have shown a synergistic effect to augment the immune response .
A discrete amount of studies are based on combination therapy with chemotherapeutic drugs, intravesical BCG, and immune checkpoint inhibitors, as some trials reported in Table 1 . Currently, an FDA-approved combination therapy is based on intravesical gemcitabine and cisplatin for NMIBCs .
As shown in Table 2, several clinical trials are designed to investigate combination therapies based on BCG immunotherapy and different chemical or biological compounds or vaccines . Combination therapies encompass also combination chemotherapies especially in recurrent and advanced BC including . Furthermore, photodynamic immunotherapy emerged recently to stimulate the immune response in NMIBC BCG-refractory or intolerant to BCG treatment as well .
One frontier of medicinal chemistry is polypharmacology . Benedetti et al. reviewed the immuno-oncological dynamic interactions to design multi-target modulators.
You May Like: Does Azo Help Bladder Spasms
Initial Patient Evaluation & Counseling
Guideline Statement 1
Prior to treatment consideration, a full history and physical exam should be performed, including an exam under anesthesia, at the time of transurethral resection of bladder tumor for a suspected invasive cancer.
Discussion
A thorough history and physical exam is important in evaluating not only bladder cancer risk but also the overall health of the patient and his/her co-morbidities. This examination will help to determine optimal management and may impact both the readiness for surgery and the type of procedure or urinary diversion that is best suited for the patient.38,39 It will also identify potential risks of surgery and identify genitourinary abnormalities that may affect pre- or intra-operative decision-making. An exam under anesthesia provides valuable information for the clinical staging and resectability of the primary tumor at surgery. This information contributes to the overall determination of clinical stage and assessment of potential benefit of neoadjuvant chemotherapy . 40,41 Presence of a large/3-dimensional, residual mass after TURBT , invasion of adjacent structures , or fixation imply locally advanced clinical stage. If the patient has hydronephrosis on imaging or on retrograde pyelogram a ureteral stent should be placed if possible to maintain or improve renal function.
Guideline Statement 2
Discussion
Guideline Statement 3
Discussion
Guideline Statement 4
Discussion
Guideline Statement 5
Neoadjuvant/Adjuvant Chemotherapy
Rationale For Use Of Neoadjuvant Treatments
Upfront chemoablative treatments like chemohyperthermia , Combat Medical Ltd., UK) and reverse thermal gel formulated with MMC are increasingly being explored as alternatives to repeat office or ambulatory procedures in patients with established, recurrent low-grade tumors. Chemoablation is a potentially attractive alternative to TURBT that may reduce treatment time, cost, and morbidity. Alternatively, others have also advocated for office-based fulguration of small, recurrent lowgrade tumors when feasible and tolerable as part of a risk-adapted strategy of treatment.30,31 Active surveillance has also been suggested as a safe and cost-effective alternative for highly selected patients.32
Upfront chemoablative treatments like chemohyperthermia and reverse thermal gel formulated with MMC are increasingly being explored as alternatives to repeat office or ambulatory procedures in patients with established, recurrent low-grade tumors.
You May Like: Loss Of Bladder Control When Running
Surgical And Technical Aspects Of Tumour Resection
5.10.2.1.Surgical strategy of resection
A complete resection, performed by either fractioned or en-bloc technique, is essential to achieve a goodprognosis .
- Piecemeal resection in fractions provides goodinformation about the vertical and horizontal extent of the tumour .
- En-bloc resection using monopolar or bipolarcurrent, Thulium-YAG or Holmium-YAG laser is feasible in selected exophytic tumours. Itprovides high-quality resected specimens with the presence of detrusor muscle in 96-100%of cases .
The technique selected is dependent on the size and location of the tumourand experience of the surgeon.
5.10.2.2.Evaluation of resection quality
The absence of detrusor muscle in the specimenis associated with a significantly higher risk of residual disease, early recurrence andtumour under-staging . The presence of detrusormuscle in the specimen is considered as the surrogate criterion of the resection qualityand is required .
It has been shown that surgical experience can improveTURB results, which supports the role of teaching programmes .Virtual training on simulators is an emerging approach . Itsrole in the teaching process still needs to be established .
5.10.2.3.Monopolar and bipolar resection
Compared to monopolar resection, bipolar resection has been introduced toreduce the risk of complications and to produce better specimens. Currently, the results remain controversial.
5.10.2.4.Office-based fulguration and laservaporisation
Tumor Recurrence And Worsening Progression Rate In Entire Patient Population
The mean age of the patients was 62.9 years and the median follow-up interval was 101.5 months . Solitary/multiple tumors were seen in 114/76 patients, respectively. Tumor recurrence occurred in 82 patients . Most patients who had tumor recurrence could be diagnosed by the routine follow-up cystoscopic examination except for 3 patients who were detected due to gross hematuria. When we divided the patients into two groups, those with or without tumor recurrence, there were no significant differences in age, gender, IVI or smoking status between the two groups . The recurrence rate in multiple tumors was significantly higher than that in solitary tumors . Univariate and multivariate analyses demonstrated that multiple tumor and absence of IVI were significant risk factors for tumor recurrence . Kaplan-Meier curves demonstrated that the 5-year recurrence free survival rate for solitary tumors was significantly higher than that for multiple tumors , and also higher for patients receiving intravesical instillation .
Table 1 Clinical characteristics of all 190 patientsFigure 2
Don’t Miss: Malignant Neoplasm Of Bladder Unspecified Icd 10
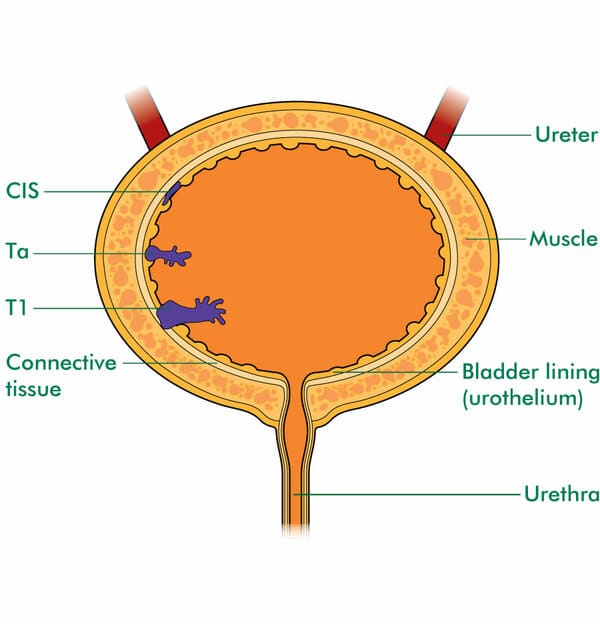
Risk Stratification Based On Clinical And Pathological Parameters
Table 1 summarizes the available predictive models to predict recurrence and progression in patients with NMIBC. The most important risk factor for progression is grade, not stage, because patients with high-grade tumors progress with similar frequency regardless of whether they were invasive or non-invasive . Millan-Rodriguez et al. evaluated a cohort of 1529 primary NMIBC patients treated with transurethral resection and random bladder biopsy and identified prognostic factors for recurrence, progression and disease-specific mortality . Multivariate analysis demonstrated that the main prognostic factors of recurrence were multiplicity, tumor size > 3 cm, presence of CIS and treatment with bacillus CalmetteGuerin . The prognostic factors for progression were grade 3 disease, multiplicity, tumor size > 3 cm, CIS and treatment with BCG. Furthermore, the prognostic factors for mortality were presence of grade 3 disease and CIS.
How Can I Prevent Bladder Cancer
You may not be able to prevent bladder cancer, but it may be helpful to know the risk factors that may increase the chance youll develop bladder cancer. Bladder cancer risk factors may include:
- Smoking cigarettes: Cigarette smoking more than doubles the risk of developing bladder cancer. Smoking pipes and cigars or being exposed to second-hand smoke also increases that risk.
- Cancer treatments: Radiation therapy is the second-most common risk factor. People who have certain chemotherapy drugs may also develop an increased risk of bladder cancer.
- Exposure to certain chemicals: People who work with chemicals, such as aromatic amines , are at an increased risk. Extensive exposure to rubber, leather, some textiles, paint and hairdressing supplies, typically related to occupational exposure, also appears to increase the risk.
- Infections: People who have frequent bladder infections, bladder stones or other urinary tract diseases may have an increased risk of developing bladder cancer.
- Past bladder cancer: People with a previous bladder cancer are at increased risk to form new or recurrent bladder tumors.
Also Check: Tcc Bladder Cancer In Dogs
Understanding The Cystoscopy Test
If your healthcare providers are concerned that you have symptoms that may be caused by bladder cancer, they will carry out a physical examination and test your urine for signs of bladder cancer. They may then use a test called cystoscopy to help make a diagnosis. This test is carried out using a cystoscope, which is a very thin, flexible tube with a tiny light and a viewing lens on the tip of the tube. It is inserted into the bladder through the urethra . This allows the healthcare provider to view the inside of the bladder and take tissue samples if needed.
Patient Stratification Into Risk Groups
To be able to facilitate treatment recommendations, the Guidelines Panelrecommends the stratification of patients into risk groups based on their probability ofprogression to muscle-invasive disease. The new risk group definitions provided in these EAUGuidelines are based on an IPD meta-analysis in primary patients and the calculation oftheir progression scores as presented in Sections 4.5 and6.1.2) .
For calculation of the risk group in individual patients,either one, or both, of the WHO 1973 and WHO 2004/2016 classification systems may be used.The probability of progression at 5 years varies from less than 1% to more than 40% betweenthe risk groups.
For factors where IPD were not collected such as variant histology, LVI,primary CIS and CIS in the prostatic urethra, literature data have been used to classifypatients into risk groups.
The clinical compositions of the new EAU NMIBC prognostic factor riskgroups based on the WHO 2004/2016 or WHO 1973 classification systems are provided in Table6.1. Apps for the web , iOS and Android are being developed to facilitate determining a patients risk groupin daily clinical practice. The individual probability of diseaseprogression at 1, 5 and 10 years for the new EAU NMIBC risk groups is presented in Table6.2.
Read Also: Muscularis Propria Invasion Bladder Cancer
C Data Abstraction And Data Management
After studies are selected for inclusion, data will be abstracted into categories that include but are not limited to: study design, year, setting, country, sample size, eligibility criteria, population and clinical characteristics , intervention characteristics , and results relevant to each key question as outlined in the previous PICOTS section. Information that will be abstracted that is relevant for assessing applicability will include the number of patients randomized relative to the number of patients enrolled, and characteristics of the population, intervention, and care settings. Sources of funding for all studies will also be recorded. All study data will be verified for accuracy and completeness by a second team member. A record of studies excluded at the full-text level with reasons for exclusion will be maintained.
Squamous Cell Carcinoma Of The Bladder

Squamous cell carcinoma is the second most common type of bladder cancer. It accounts for about 5 percent of bladder cancers in North America and Europe. This cancer begins in the thin, flat squamous cells that may form in the bladder after chronic inflammation and infection. Squamous cell carcinoma is most often found in parts of the world where a parasitic infection called schistosomiasis is widespread, such as the Middle East.
Also Check: Does Hair Dye Cause Bladder Cancer
Risk Groups For Non Muscle Invasive Bladder Cancer
Doctors put non muscle invasive bladder cancer into 3 risk groups. These groups describe how likely it is that your cancer will spread further, or come back after treatment.
The 3 risk groups are:
- intermediate risk
Your doctor tells you whether your cancer is low risk, intermediate risk or high risk. Knowing your risk group helps them decide which tests and treatment are best for you.
Your risk group depends on:
- the size of your tumour
- what the cells look like under a microscope
- how many tumours there are
- the type of bladder tumour
- whether you have had treatment in the last year for early bladder cancer
Health Economics Of Nmibc
NMIBC is one of the most costly cancers to manage on a per-patient basis because of its high prevalence, high recurrence rate, need for adjuvant treatments and the requirement for long-term cystoscopic surveillance. The total cost of treatment and 5-year follow-up of patients with NMIBC diagnosed in the UK has increased from £73million to £213million from 2001 to 2012 . From a patient perspective, there often are considerable anxieties about recurrences, transurethral resection and progression, requiring additional therapies with potential mortality and long-term morbidity . Transurethral resection itself is associated with reduced quality of life, including both mental and physical health domains, although these effects are usually transient. Substantial effects on health-related quality of life are most likely to come from adjuvant intravesical treatments and radical or palliative treatments for progression. The cost-effectiveness of NMIBC treatment strategies has not been widely studied.
Recommended Reading: What Is The Treatment For Low Grade Bladder Cancer
Rare Forms Of Bladder Cancer
Adenocarcinomas account for less than 2% of primary bladder tumors. These lesions are observed most commonly in exstrophic bladders and are often associated with malignant degeneration of a persistent urachal remnant.
Other rare forms of bladder cancer include leiomyosarcoma, rhabdosarcoma, carcinosarcoma, lymphoma, and small cell carcinoma. Leiomyosarcoma is the most common sarcoma of the bladder. Rhabdomyosarcomas most commonly occur in children. Carcinosarcomas are highly malignant tumors that contain a combination of mesenchymal and epithelial elements. Primary bladder lymphomas arise in the submucosa of the bladder. Except for lymphomas, all these rare bladder cancers carry a poor prognosis.
Small cell carcinoma of the urinary bladder is a poorly differentiated, malignant neoplasm that originates from urothelial stem cells and has variable expression of neuroendocrine markers. Morphologically, it shares features of small cell carcinoma of other organs, including the lung.
Section : Clinical Concerns Related To The Intravesical Chemotherapy
Question 7: What are the contraindications to immediate postoperative intravesical chemotherapy?
Recommendation: Immediate post-operative intravesical chemotherapy is contraindicated in the patients with suspected bladder perforation or severe hematuria. .
Evidence summary: We referred to the recommendations from the EAU guideline , AUA guideline , CUA guideline , NCCN guideline , and Guidelines for Diagnosis and Treatment of Urology and Andrology in China .
Question 8: Does immediate postoperative intravesical chemotherapy reduce the risk of recurrence of NMIBC?
Recommendation: Except for those with the contraindications to immediate intravesical chemotherapy after operation, all patients with NMIBC should receive immediate postoperative intravesical chemotherapy within 24 h after TURBT. .
Question 9: Do patients with low-risk tumors only need SIC after TURBT?
Recommendation: Only SIC following TURBT is needed for patients with low-risk tumors. .
Evidence summary: We referred to the recommendations from the EAU guideline , AUA guideline , CUA guideline , NICE guideline , NCCN guideline , and Guidelines for Diagnosis and Treatment of Urology and Andrology in China, summary of recommendations on chemotherapy for NMIBC patients with different risk levels are shown in Additional file : Table S1 .
Question 10: What are the commonly used drugs and doses for the intravesical chemotherapy?
Question 11: How to improve the efficacy of intravesical chemotherapy?
Don’t Miss: Rare Types Of Bladder Cancer
A Randomized Trial Of Photodynamic Surgery In Nonmuscle
- Rakesh Heer, F.R.C.S.1,
- James NDow, F.R.C.S.12,
- John Norrie, M.Sc.13,
- Graeme MacLennan, M.Sc.3, and
- Emma Hall, Ph.D.2
- 1Newcastle University, Newcastle upon Tyne, United Kingdom
- 2The Institute of Cancer Research, London
- 3Centre for Healthcare Randomised Trials, University of Aberdeen, Aberdeen, United Kingdom
- 4Edinburgh Bladder Cancer Surgery, Department of Urology, Western General Hospital, Edinburgh
- 5South Tees Hospitals NHS Trust, Middlesbrough, United Kingdom
- 6Royal Devon and Exeter Hospital NHS Trust, Exeter, United Kingdom
- 7University of Dundee, Dundee, United Kingdom
- 8Basingstoke and North Hampshire NHS Foundation Trust, Basingstoke, United Kingdom
- 9Royal Liverpool and Broadgreen University Hospitals NHS Trust, Liverpool, United Kingdom
- 10University College London Hospitals NHS Foundation Trust, London
- 11PHOTO Trial patient representative, United Kingdom
- 12Academic Urology Unit, University of Aberdeen, Aberdeen, United Kingdom
- 13Edinburgh Clinical Trials Unit, Edinburgh University, Edinburgh
- 14Health Services Research Unit, University of Aberdeen, Aberdeen, United Kingdom
Types Of Adjuvant Therapy
Adjuvant treatment is any agent administered after complete TURBT aimed at reducing the risks of NMIBC recurrence and progression. Traditionally, only intravesical agents have been used in this setting, but emerging data on the use of systemic checkpoint inhibitor immunotherapies either as a monotherapy or combined with intravesical agents may radically change this treatment paradigm. Current adjuvant intravesical treatments can be defined by the timing and sequence of their administration: perioperatively, usually within 24h of TURBT, induction, as an initial course for that particular agent, or maintenance, subsequent courses in the setting of no evidence of disease after TURBT and induction treatment.
Standard intravesical therapies can also be categorized as either chemotherapy or immunotherapy agents. The most used contemporary chemotherapy agents are mitomycin C, gemcitabine, epirubicin, and docetaxel. These are given individually or in combination depending on the indication. Historically, agents such as doxorubicin, valrubicin, cisplatin, and thiotepa, among others, were also frequently studied and used. The gold standard intravesical immunotherapy is BCG, an attenuated mycobacterium strain first described for use in bladder cancer in 1976. Interferon has also been studied, but contemporary use is limited.
Read Also: My Bladder Is Always Full