Postoperative Monitoring And Adjustments
After surgery each dog received injectable opiate analgesics at standard dosage and intervals for at least 48 hours, combined with nonsteroidal drugs or other analgesics for 7 days. On the 2nd postoperative day, the bladder of each dog was catheterized with the dual lumen catheter to allow bladder pressure responses to externally driven nerve stimulation to be recorded . The bladder pressures in response to electrical sacral spinal nerve root stimulation were recorded and the stimulation parameters on the external stimulating device were varied to achieve an âonâ/âoffâ stimulus pattern that generated sufficient bladder pressure to empty the bladder to an appropriate residual volume while avoiding excessive intravesicular pressure . Four factors were available for altering the stimulus parameters: amplitude of stimulation, pulse width, pulse repetition frequency, and the total duration of stimulating and relaxing epochs.
The owners were asked to return dogs after 3 weeks and to inform the clinic of any problems in the meantime. Subsequent follow-up was by telephone or visits as required by each owner and dog. Urine of dogs in which there was evidence of urinary tract infection was routinely analyzed , cultured for bacterial growth and antibiotic sensitivity as required, and treatment with appropriate antibiotics administered for 3 weeks when needed.
What Is Overactive Bladder
Millions of people suffer from a bladder control problem known as overactive bladder. This condition involves the need to urinate frequently both day and night. While there are many treatment options available: antimuscarinic medications, behavior / dietary modifications, vaginal estrogen, and pelvic floor exercises, not all patients respond well to these therapies. InterStim is a third-line therapy used to control overactive bladder symptoms with an implanted device that stimulates the sacral nerve.
How Is An Interstim Device Implanted
InterStim placement is quickly and safely done at an outpatient surgery center. The procedure itself typically takes between 20-30 minutes to complete, and surgery is minimally invasive. This can often be done under local anesthesia, though some doctors will prefer to use general anesthesia. The device is inserted in the lower back, above the buttocks.
Though insurance plans vary, Medicare and private insurance generally cover InterStim treatment, but please consult with your doctor and insurance company.
You May Like: Does Prostate Cancer Spread To Bladder
What Is The Outlook
You may need to try different treatments until you have relief of your symptoms. It is important to know that none of the treatments usually works right away. It can often take weeks to even months before you notice an improvement in your symptoms. Even with successful treatment, your IC/PBS may not be completely cured.
However, most patients can have significant relief of their symptoms and lead a normal life with the right treatment. You may find that you still experience some symptoms, however, and may find that you constantly have to pass urine more frequently. It is likely that you will always have to avoid certain types of food that have made your symptoms worse in the past.
You May Like: Cure A Bladder Infection In As Little As 6 Hours
The Solution: A Bladder Stimulator
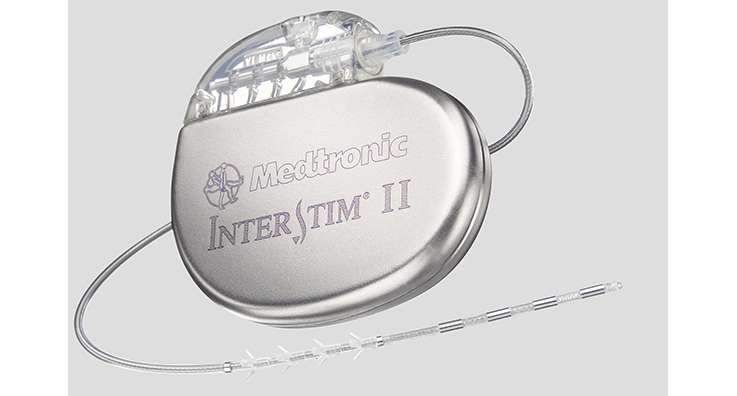
A bladder stimulator is a small device implanted in your back at the base of your spine and above the buttocks during a procedure known as sacral nerve stimulation . The sacral nerves carry the signals between your bladder, spinal cord, and brain that tell you when you need to urinate. This is done after exhausting conventional treatment options such as pelvic floor physical therapy and medications.
At GSMC, we use a long-term relief technology called Axonics Therapy. Before committing to long-term therapy, our team first does a trial with the patient to see if the treatment works for each and every patient. A thin thin wire is carefully placed close to the third sacral nerve through a needle. The wire connects to a battery-powered device called a pulse generator that you wear outside your body during a three day trial period. If the symptoms get significantly better the device is put in permanently and can last up to 15 years.
The actual electrical stimulation that is used to treat urinary incontinence sends a mild electric current to nerves in the lower back/pelvic muscles which are involved in urination. The patient does not feel this electrical stimulation but the nerves do leading to symptoms improvement.
Think you may be a candidate for a bladder stimulator? Call to set up your consultation and begin the recovery process today: 732-202-3000
Also Check: Can Soda Cause Bladder Infection
Sacral Nerve Stimulation For Incontinence
Sacral nerve stimulation involves the placement of an implant to stimulate the sacral nerve.
Sacral nerve stimulation , also called sacral neuromodulation, involves surgical implantation of a device that sends a low-voltage electrical current to the sacral nerve. The sacral nerve is located at the base of the spine that affects the bladder, bowel, and pelvic floor. The implant stimulates the sacral nerve and to alleviate fecal and/or urinary incontinence. A hand-held device is used to stop the contraction of your sphincter muscles when you need to empty your bowels.
Who Benefits from Sacral Nerve Stimulation
Adult patients experiencing urinary incontinence due to retention and/or overactive bladder with urge incontinence, and individuals with fecal incontinence and/or chronic constipation who have failed other treatments may be able to try this treatment. You must be able to operate the hand-held programmer device. There are medical conditions that may prevent the safe use of this sacral nerve stimulator. Also if you need certain medical tests, you may not be able to use this treatment. Ask your doctor if you are a possible candidate.
If you have bowel incontinence or chronic constipation, use of the sacral nerve stimulator may result in improvement in your ability to delay emptying your bowels, decrease the number of episodes of bowel incontinence, and improve your quality of life.
Things to Keep in Mind
Medical Reviewer: Carrie Carls , RN, BSN, CWOCN, CHRN
Devices Available For Pudendal Nerve Stimulation
The pudendal nerve can be stimulated on a chronic basis using a tined quadripolar lead or an implant-able microstimulator . Neither of these applications is FDA approved for pudendal stimulation. The development of a tined lead for sacral nerve stimulation allows implantation of the lead without suture fixation . This feature makes it possible to deploy the lead at other sites, such as the pudendal nerve. The advantage is that the lead can be placed at the pudendal nerve by means of a posterior approach, externalized, and tested on an outpatient basis to assess the clinical response. If the patient demonstrates significant improvement in their voiding symptoms, a permanent IPG can be placed in a subcutaneous pocket in the upper buttock.
The Bion microstimulator is a miniature, self-contained, rechargeable, implantable neurostimulator that is designed as a platform technology and is intended to treat a wide variety of disorders through direct electrical stimulation. This device is approved in Europe for commercial use in pudendal nerve stimulation and is in clinical trials in the United States for urinary urge incontinence, urgency or frequency, and chronic headache. The fully integrated microstimulator contains a rechargeable battery, a radio transmitter, an antenna for bidirectional telemetry, a programmable microchip, and stimulating electrode. The implant is 28 mm long and 3.2 mm in diameter, and it has a mass of only 0.7 g .
Andrew Ruys, in, 2019
Read Also: Does Bladder Cancer Always Come Back
Can Bladder Endometriosis Be Treated By Minimally Invasive Techniques
Yes, but only by an experienced surgeon. To resect a portion of the bladder and repair the bladder by video-assisted laparoscopy or robotic-assisted laparoscopy requires a high level of skill with those instruments. As mentioned, Drs. Nezhat were the first to perform minimally invasive surgeries for the treatment of all forms of endometriosis, including bladder endometriosis. After nearly 3 decades, Drs. Nezhat have now performed among the most, if not the most, endometriosis surgeries in the world using minimally invasive and robotic techniques.
A cystoscopy, as shown above, is a procedure in which a camera is placed inside of your bladder to help your doctor visualize your bladder and provide an accurate diagnosis.
However, even a cystoscopy can sometimes miss endometriosis that affects only the outside of the bladder. Therefore, multiple diagnostic tools are sometimes required to achieve an accurate diagnosis.
With the bladder so close to the uterus, its sometimes difficult for patients to determine from which organ the pain is emanating.
Thats why a careful examination is required so that your doctor can properly treat each area.
A common symptom of bladder endometriosis is urinary frequency. However, urinary frequency can also be a symptom of many other disorders.
Thats why its important that you receive a very careful examination to rule out other disorders.
Read Also: Homeopathic Cure For Bladder Infection
Stimulation Of Sacral Nerves And Sacral Roots For Continence
During the 1980s, Tanagho and Schmidt developed the use of electrical stimulation of sacral nerves to treat a variety of lower urinary tract problems, including those of the neurogenic bladder . Subsequently, a neuroprosthesis was developed which comprised an implanted pulse generator attached to a multipole electrode surgically inserted into the S3 sacral foramina for stimulating the mixed sacral nerves . This is marketed as Interstim by Medtronic, Inc., of Minneapolis, U.S.A., and is in clinical use for patients without SCI, some of whom gain relief of symptoms of an overactive bladder and improved continence . The device has occasionally been used in patients with an SCI with variable results and is not FDA approved for this condition.
In a recent pilot study similar benefits have been shown using a Finetech-Brindley implant to stimulate extradural or intradural sacral roots . In a small group of patients with a suprasacral spinal injury, electrodes were placed bilaterally on either the mixed extradural sacral roots or separated anterior and posterior sacral roots intrathecally without deafferentation. Each patient was assessed preoperatively with DPN stimulation, as described above, to demonstrate the efficacy of neuromodulation. Preliminary results indicated that patients were able to achieve both good suppression of detrusor hyperreflexia and clinically useful increases in bladder volume
Matthew R. Cooperberg, Marshall L. Stoller, in, 2008
Recommended Reading: I Have A Weak Bladder Help
Who Gets Interstitial Cystitis
As many as 90% of people with IC are women. Somewhere between about 3% to 6% of adult women have some form of IC. Thatâs about 3 million to 8 million American women. About 1.3% of American men also have it.
On average, people first start having problems in their 40s. The risk of getting it goes up as you get older.
How Does Sacral Nerve Stimulation Actually Work
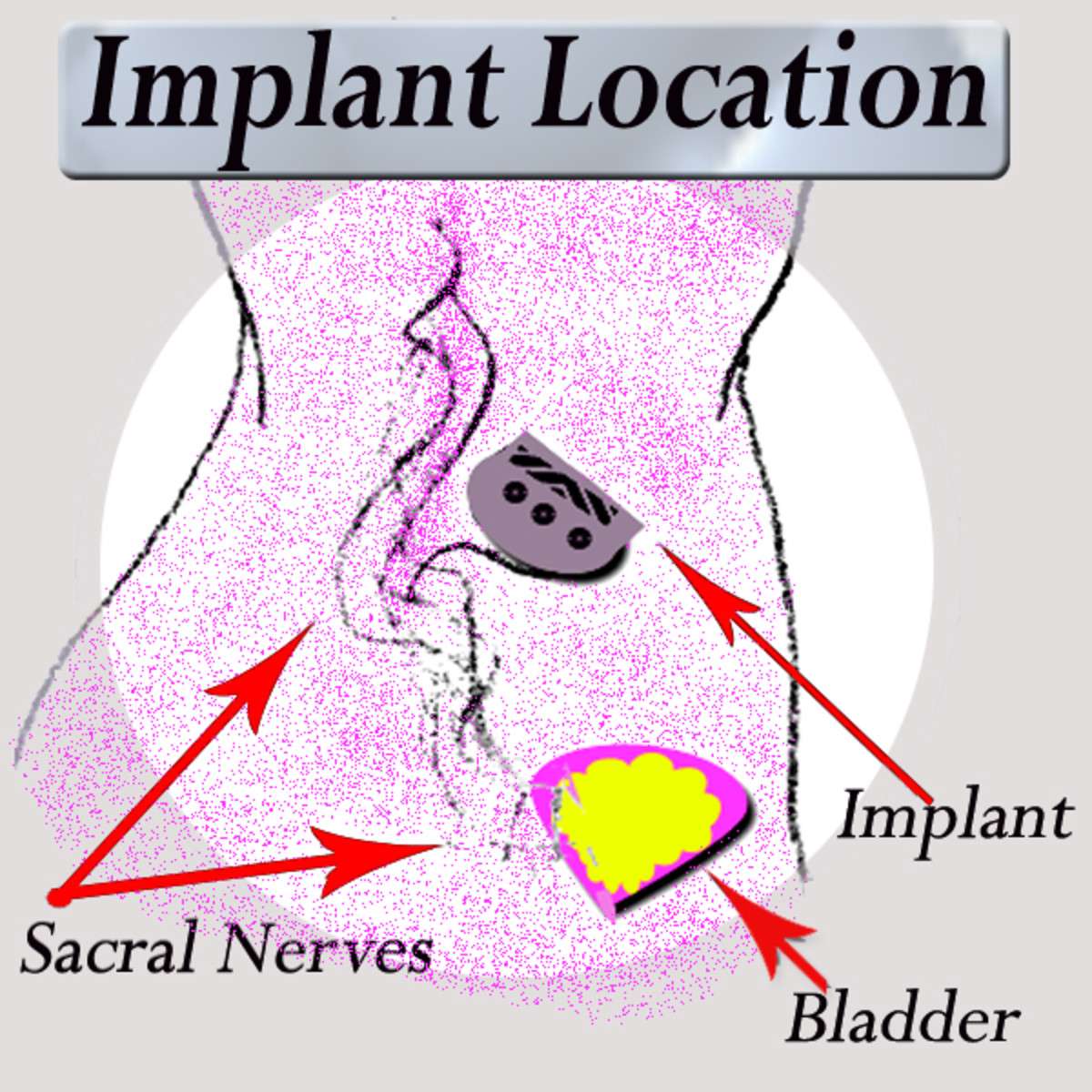
Sacral nerve stimulation therapy uses a small device that is implanted under the skin in the upper buttock area. The device sends mild electrical impulses through a lead that is positioned close to a nervelocated in the lower back , which influences the bladder,the sphincter and the pelvic floor muscles.
A neurotransmitter device, implanted under the skin in the upper buttock area, transmits mild electrical impulses through a lead wire close to the sacral nerve. The impulses, in turn, influence the bladder sphincter and pelvic floor muscles providing bladder control.
You May Like: Treatment Of Overactive Bladder In Males
What Does The Procedure Involve
This procedure involves an initial test phase to stimulate the nerves that control some aspects of bladder function these nerves are situated in front of the sacrum . If the test is successful, at a later date a permanent lead is placed into the sacrum and attached to a permanent nerve stimulator which is inserted into the buttock area.
Staged Implant Retrospective Study
This was a retrospective study conducted in patients who had inconclusive test stimulation results with the Medtronic model 3057 test stimulation lead. In this study, the test stimulation typically lasted for 3-5 days, but no more than 7 days. Participating physicians reported that 76 of the 80 patients had a successful screening period with the staged implant. Of the 80 patients screened, 73 went on to undergo Interstim implantation, 60 of whom were noted to have a successful outcome during an average follow-up of 1.6 years . Of the 80 patients who were screened with a long-term lead for a staged implant, 47 had also been screened with a temporary model 3057 test stimulation lead.
Test stimulation was unsuccessful with the model 2057 lead in 24 of the 47 patients prior to receiving the staged implant. Screening with the long-term lead was successful in 21 of these 24 patients. Thus, it appears that use of the long-term lead for screening as part of a staged implant may be helpful in patients with inconclusive results from screening with the model 3057 lead, to determine if they will benefit from Interstim implantation.
Don’t Miss: Medicine To Stop Bladder Spasms
What Is Sacral Neuromodulation
Sacral neuromodulation, also known as sacral nerve stimulator, is a procedure where a bladder stimulator is implanted for urinary control that targets the communication issue between the brain and nerves that control the bladder. Sacral neuromodulation is used for treating urge incontinence and symptoms of urgency-frequency. Sacral neuromodulation involves the placement of a small device in the body. This device modulates the sacral nerves with mild electrical pulses, helping the brain and the nerves to communicate so the bladder can function properly.
Interstim Therapy For Urinary Incontinence
Urinary incontinence affects over 25 million Americans. A number of causes exist: injury or trauma to the pelvic region or back, enlarged prostate or prostate cancer, pregnancy or childbirth, neurological disorders, estrogen loss following menopause, other health conditions like multiple sclerosis or suffering a stroke.
Take our Urinary Incontinence Self-Assessment
Also Check: What Kind Of Doctor Treats Bladder Problems
Sacral Nerve And Pelvic Floor Muscle Stimulation Using External Interferential Stimulators
In the mid 1990s, external interferential stimulators have become available that appear to provide the same pattern of sacral nerve stimulation and pelvic floor relaxation as the implanted Interstim devices without the need for surgery or the attendant complications. These devices often confer pelvic floor relaxation and relief of urinary urgency and/or prostatic pain within a week of use and can eventually prevent exercise-induced pain and dyspareunia. Studies of similar devices in specific conditions such as overactive bladder syndrome have shown that these have been significantly more effective than treatment with medications such as oxybutinin.
With proper adjustment and programming, externally placed interferential stimulators can directly stimulate sacral nerve roots, causing pelvic floor relaxation. With enough intensity, this stimulation could transiently paralyze pelvic floor muscles leading to incontinence and, someday soon, to the development of new devices such as a noninvasive Foley catheter and electronic laxatives. As is the case with implanted sacral nerve stimulators, interferential stimulators appear to function better with prolonged use. This could be due to the fact that repeated stimulation of a dystonic or chronically spasmodic area of muscle will reduce muscular edema and improve venous outflow.
Christopher Kennerly Payne, in, 2008
Interstim For Overactive Bladder
InterStim is a therapy for OAB. Many women find medications are ineffective against the symptoms of OAB. If medications didnt work for you or if side effects were impacting your quality of life, InterStim may be an option. An InterStim device can reduce OAB symptoms by that restoring normal communication between the brain and the bladder and the bowel.
How does InterStim for OAB work?InterStim uses sacral nerve stimulation therapy, or sacral neuromodulation, to treat OAB. The Medtronic InterStim an FDA-approved sacral nerve stimulation device. A small device is implanted that sends electrical impulses to nerves in the lower back. These nerves are called the sacral nerves. They are stimulated gently to decrease the symptoms of OAB.
For two weeks, you will use a temporary InterStim device. The temporary device is the same as a permanent device, but it is not surgically implanted. Bladder and bowel functions and improvements are recorded in a journal during the trial. If the trial goes well, youll undergo surgery for permanent InterStim placement.
Once a permanent device is installed, you can use a remote to increase or decrease the nerve stimulation to adjust bladder function. InterStim may reduce or completely eliminate your OAB symptoms.
What results can I expect?Studies show that InterStim is safe and effective. InterStim is proven to restore bladder function, return bladder control long-term, and improve quality of life.
Recommended Reading: Depend Adult Bladder Control Pads
Who Is Not Suitable For Treatment With Sacral Neuromodulation
InterStim® therapy is not recommended for usage in:
- People in whom the stage 1 trial or test stimulation was unsuccessful
- People who are unable to properly operate the hand held patient programmer
- People with urinary blockage
- Movement or migration of the pacing lead
- Mechanical problems with the device
- Interactions with other devices or diagnostic equipment such as MRI
- Changes in urinary or bowel function which are undesirable
Most of these problems can be resolved by changes in programming parameters of the InterStim® device.
Some patients with the permanent InterStim® device need reoperation before the pacemaker battery needs replacement due to:
- Reduced or loss of effectiveness of the InterStim® device
- Pain at the lead or pacemaker site
- Infection of the device
It is thought that reoperation rates are reducing over time with refinements in the InterStim® device implantation techniques and equipment.
Dr. Karen McKertich
Read Also: Natural Remedies For Cystitis Bladder Infection
Risks Of Sacral Nerve Stimulation
With any surgery there are risks. The risks for nerve stimulation are related to the device or the procedure to implant the device. The main risks include infection related to the device or mechanical failure where the device would need to be reprogrammed or revised.
Because the neurotransmitter can be removed at any time, there are few risks associated with sacral nerve stimulation. However, you should never undergo any kind of heat treatment for sore muscles when the neurotransmitter is implanted or it could cause severe injury. Some other side effects may include:
- Technical problems with the neurotransmitter
- Infection, skin erosion or pain at site of implant
- Tickling, numbness or burning
- Migration of the device under the skin
Don’t Miss: Bcg Used In Bladder Cancer
Finally A Diagnosis Of Interstitial Cystitis
Back in New Zealand, I found a pelvic floor physiotherapist. I realised through this therapy, that my pelvic floor was clenched on all of the time, creating more issues. With the therapy I learned to relax those muscles which, in turn, decreased some of the pain. After completing a second cystoscopy with yet another urologist, I was diagnosed with Interstitial cystitis with Hunners ulcers. I was so excited to have a name for this condition. Finally!!! This was good news. The bad news? There was no cure for Interstitial Cystitis.