What I Need To Know About Sacral Nerve Stimulation
Sacral nerve stimulation is treatment for urinary retention without blockage, overactive bladder symptoms, and fecal incontinence. Overactive bladder symptoms include urinary urge incontinence and urinary frequency. Electrical impulses are sent directly to the sacral nerves to improve or restore bladder or bowel function. Sacral nerves are in your lower back. They control anus, rectum, and bladder functioning. SNS is done when medicine and behavior therapy do not help improve symptoms. Implantation of an SNS includes 2 stages. The first stage is a trial phase to see if SNS will improve your symptoms. The second stage involves implanting the stimulator.
Is It Covered By Insurance
Many insurance companies cover sacral nerve stimulation in people who do not respond to more conservative treatments or who do not make good candidates for other treatments.
For example, Medicare plans supported by Blue Cross North Carolina cover sacral nerve stimulation when used to treat urinary urge incontinence. To qualify, you must show a 50 percent improvement in the evaluation phase and meet other candidacy requirements.
Sacral nerve stimulation is a procedure that involves placing an electrode under your skin to stimulate the nerves that send messages between your brain and bladder.
It may be an effective option if more conservative treatments have failed. You can discuss this procedure with a doctor to see if itâs right for you.
Before receiving an implant, youâll be given the opportunity to have a short trial to see if itâs effective.
Radiofrequency Therapy The Latest Technology
Abnormal electrical activity may start in one area of the bladder then spread across the organ, causing OAB symptoms, Dr. Goldman explains. Two companies are developing technologies designed to disrupt or contain that activity.
NewUros transurethral bladder partitioning device uses controlled radiofrequency energy to ablate thin tissue lines to fence off abnormal electrical activity in the bladder. NewUros TBP has a disposable radiofrequency catheter that is inserted through the urethra and expanded like a balloon so it is in contact with the bladder wall. A radiofrequency generator sends power to the catheter, creating thin radiofrequency ablation lines that partition the bladder into electrically independent zones. This device has not yet undergone human clinical trials.
Amphora Medical is developing a cystoscopic device capable of ablating areas near the trigone an area in the bladder where it is thought many of the nerves that control function pass through with radiofrequency energy. This could dampen nerve signals and restore normal bladder function. The company is currently running two clinical trials, one in Belgium and Canada and one in the U.S.
Also Check: Is Bladder Leakage Common After Hysterectomy
What This Means For Oab Treatment Questions Remain
The mechanism of action in SNM has long been hypothesized to involve influencing neural circuits in the peripheral and central nervous system . This study showed real-time changes in brain activity with SNM therapy, supporting the hypothesis that SNM works in part by influencing pathways in the CNS. We also observed that altered stimulus intensity elicited an amplitude-response relationship in various brain regions.
Our challenge now is to further clarify how changes in brain activity from SNM affect urinary tract function. Does SNM provide a modulatory signal that alters the set point in brain centers that coordinate lower urinary tract function and voiding like a thermostat? In OAB, this set point is likely miscalibrated.
Or does SNM produce afferent activity that alters aberrant neural activity underlying OAB? An analogy for this is how noise-cancellation headphones enable soft music to be heard in a noisy environment.
Our study achieved its aim of showing that SNM devices alter brain activity. In the future, we hope to change the parameters used in fMRI testing, such as performing fMRI when patients have a full bladder or with different stimulus settings, to tease out more details on how SNM devices affect neural structures and circuits.
What Is Sacral Neuromodulation
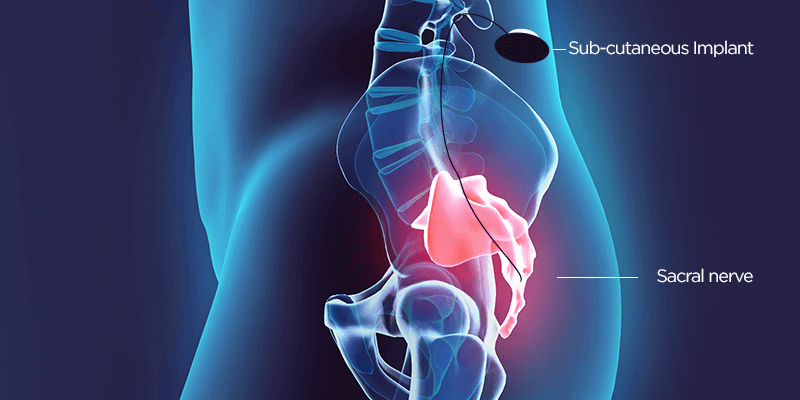
Neuromodulation therapy involves the delivery of tiny electrical impulses to nerves in order to change how they work. Sacral neuromodulation works by changing how the sacral nerves work. These nerves carry messages between the spinal cord and the bladder and their nerve signals are malfunctioning in OAB. Sacral neuromodulation interrupts the abnormal signals and helps to improve OAB symptoms.
You May Like: Botox Injection For Bladder Incontinence
How Does Sacral Nerve Stimulation Actually Work
Sacral nerve stimulation therapy uses a small device that is implanted under the skin in the upper buttock area. The device sends mild electrical impulses through a lead that is positioned close to a nervelocated in the lower back , which influences the bladder,the sphincter and the pelvic floor muscles.
A neurotransmitter device, implanted under the skin in the upper buttock area, transmits mild electrical impulses through a lead wire close to the sacral nerve. The impulses, in turn, influence the bladder sphincter and pelvic floor muscles providing bladder control.
How Successful Are The Latest Third
Cleveland Clinic physicians recently published a review article in the Current Urology Reports that evaluates new OAB devices and technologies.
Some of these new technologies are simply improvements on methods already in use for controlling OAB symptoms, while others offer a whole new way of attacking the problem, reports urologist Howard Goldman, MD, one of the study co-authors. Their review is summarized below.
Read Also: Antibiotics For Bladder Infection While Pregnant
Improvements To Tibial Nerve Stimulation
Another OAB treatment modality included in the recent review is percutaneous tibial nerve stimulation . Physicians insert a small needle into the ankle to stimulate the tibial nerve once a week for the first three months, followed by ongoing monthly appointments .
Tibial nerve stimulation for OAB.
Two new implantable TNS devices have potential to eliminate the need for the frequent office visits. One is the Bioness StimRouterTM neuromodulation system. It has an implanted lead with an integrated receiver, anchor and three electrode contacts in close association to the posterior tibial nerve. The lead captures stimulation energy that is delivered wirelessly and transdermally from a rechargeable external pulse transmitter and electrode patch. Both the transmitter and patch are worn only during periods of stimulation. The patient uses a programmer to track usage and change programs. The programmer controls the external pulse transmitter wirelessly. Dr. Goldman is the site principal investigator for an ongoing clinical trial of this technology.
BlueWind Medicals RENOVATM TNS device can be surgically implanted or percutaneously injected in close vicinity to the tibial neurovascular bundle. Powered through wireless technology, this device includes an external control unit worn around the ankle during treatment 30 minutes daily. Using a computer program, the physician sets stimulation parameters for each patient to optimize therapeutic outcome.
What Are The Benefits And Risks Of Sacral Nerve Stimulation Therapy
The benefits of nerve stimulation therapy are that it can greatly reduce or eliminate bladder control problems, specifically urinary urge incontinence and urgency-frequency symptoms, in people who have symptoms of overactive bladder. It is also a type of treatment that can be completely reversed and discontinued at any time without permanent damage to the nerves.
Most risks or side effects are related to the device or the implantation procedure itself and are low and/or uncommon. The main risks include infection , related to device implantation, or mechanical failure, in which case, the device would need to be reprogrammed or revised. Other problems such as pain at the implant site, lead movement, technical problems, and undesirable stimulations/sensations are not common and can be resolved, says the device manufacturer.
Also Check: Is Bladder Cancer Slow Growing
How Does It Feel
The implant is very small and placed underneath the fat of the lower back/upper buttock area. The surgeon makes a small incision of about 3cm to insert the device during surgery, so recovery is quick and straightforward. The device is cushioned by the bodys fat and so you will not be aware that you have any implant in that area. Patients say they are sometimes aware of the device when they lie in certain positions, but they do not report feeling any discomfort.
The electrical stimulation is kept below detectable levels and the patient is not aware of the stimulation unless the pulses are turned up. There is usually an initial period of the first few weeks after the implant is inserted when you get used to the device and find the right level for you.
Data Collected With Snm On And Off
For the study, we turned off patients SNM devices for one week, causing their symptoms to return. Then we performed fMRI on each patient while we turned the SNM on and off. Patients underwent three fMRI sessions with their devices operating at different levels of intensity each time: subsensory, sensory and suprasensory.
FMRI images demonstrating areas of deactivation and activation in the brain at different intensities of stimulation.
The data yielded some interesting results. In general, brain activation progressed with increased stimulation levels. However, activation of the inferior frontal gyrus remained stable, and deactivation of the pons and the periaqueductal gray matter only occurred with subsensory stimulation. Sensory stimulation activated the insula, which previous studies have shown is activated upon bladder filling, but deactivated the medial and superior parietal lobes. Suprasensory stimulation activated multiple structures, including sensory regions of the S3 nerve root where the stimulator lead is implanted.
Also Check: Unable To Control Bowels And Bladder
When To See A Health Care Provider And What To Expect
Talk to your health care provider if you have urinary incontinence or any signs of a bladder problem, such as:
- Needing to urinate more frequently or suddenly
- Cloudy urine
- Urinating eight or more times in one day
- Passing only small amounts of urine after strong urges to urinate
- Trouble starting or having a weak stream while urinating
Your doctor may recommend urodynamic testing and perform the following to try to figure out what might be causing your bladder problem:
- Give you a physical exam and take your medical history.
- Ask about your symptoms and the medications you take.
- Take urine and blood samples.
- Examine the inside of your bladder using a cystoscope a long, thin tube that slides up into the bladder through the urethra. This is usually done by a urinary specialist.
- Fill the bladder with warm fluid and use a cystoscope to check how much fluid your bladder can hold before leaking.
- Order or perform a bladder ultrasound to see if you are fully emptying your bladder with each void.
- Ask you to keep a daily diary of when you urinate and when you leak urine. Your primary care doctor may also send you to a urologist, a doctor who specializes in urinary tract problems.
What Is Involved In The Trial Stage Or Stage I Interstim Implant

The trial stage or Stage 1 InterStim® implant involves a day case procedure under anaesthesia with insertion of a fine wire through the skin over the tailbone which is positioned next to one of the sacral nerves. The fine wire is taped to the skin and covered by surgical dressings so that it is not disturbed for the duration of the trial stage. The wire is connected to the external controller that is worn like a paging device under the clothing for the trial period.
The duration of the trial phase is usually 10 to 14 days. The final pacemaker device is NOT implanted during the trial phase and hence the InterStim® device is controlled during the trial phase by a connection to a temporary external device.
Patients are taught to use the external controller and keep a bladder diary during the trial stage to assess if there is a significant and worthwhile improvement in OAB symptoms.
Read Also: Bladder Cancer That Has Metastasized
What Does Treatment With Sacral Neuromodulation Involve
The InterStim® device works like a pacemaker for the bladder in a similar way that a cardiac pacemaker works for the heart.
Surgery to insert the InterStim® device involves a two staged procedure.
Stage 1 the trial stage involves the insertion of a pacing wire alongside a sacral nerve using X-ray guidance as a day case procedure.
- If the trial period is successful in reducing urinary symptoms of bladder overactivity by at least 50%, the patient can proceed to implantation of the full pacemaker device.
- The advantage of the trial stage is the ability to test out the InterStim® device over several days to see if it suits the individual before committing to the treatment.
- About 7 in 10 patients choose to move from the test stimulation to have the permanent InterStim® pacemaker inserted.
Images supplied courtesy of Medtronic Inc.
Stage 2 the insertion of the permanent InterStim® pacemaker involves the implantation of a device smaller than the size of a box of matches into the fat of the buttock where it cannot be seen.
Images supplied courtesy of Medtronic Inc.
What Is Axonics Sacral Neuromodulation Therapy
Sacral neuromodulation therapy, also known as sacral neuromodulation therapy for urinary retention, is a clinically-proven, FDA-approved treatment to reduce your symptoms. The therapy, given through an implant, is also a treatment for urinary urgency incontinence. There is also sacral neuromodulation for bowel control, helping to stop or limit an overactive bowel, and sacral neuromodulation for urinary retention.
Sacral neuromodulation therapy works by conducting sacral nerve stimulation using an implanted device to help your brain, bladder, and bowel communicate. With better communication, your body will once again work together, preventing accidents and false signals.
Sacral neuromodulation therapy comes with many benefits. The treatment is known to be safe, clinically-proven, and long-lasting. The Axonics sacral neuromodulation therapy device is built to give you 15 years of relief from your overactive bladder. The procedure is outpatient, allowing you to return home the same or next day. Sacral nerve stimulation surgery recovery will take several weeks and possibly up to two months to completely heal.
Discomfort from the device is rare. Less than 2% of patients experience any discomfort at the implant site . Sacral nerve stimulation side effects will be explained by your physician prior to the procedure. If you choose to move forward with the procedure, sacral nerve stimulation after care will also be discussed.
Also Check: How To Cope With Overactive Bladder
What Happens Before Sacral Nerve Stimulation
Before the device is implanted, you must undergo a trial assessment period called a Peripheral Nerve Evaluation where a test simulation electrode is implanted. In an outpatient procedure, your doctor will use local anesthetic to numb the area and place a temporary device. This test simulation is a one to two-week trial that allows you and your doctor to make an informed decision about the long-term value of sacral nerve stimulation.
The trial will demonstrate the effects of electrical impulse on your bladder and bowel control system during everyday activities. If the trial shows improvement in urine and fecal leakage, urgency or frequency by at least half, you may proceed with the implantation of the long-term device.
How Does Sacral Neuromodulation Work
The most widely studied form of sacral neuromodulation uses the InterStim® device . InterStim® works by delivering an electrical message to the sacral nerves which modify abnormal reflexes in the sacral and pelvic nerves that supply the bladder and pelvic floor, as well as modifying abnormal communications between the brain and bladder.
These abnormal reflexes may cause problems with:
- An overactive bladder with frequency, urgency and urge urinary incontinence as well as
- Some cases of urinary retention or inability to empty the bladder that are NOT related to blockage in the urinary tract
Recommended Reading: Why Can I Control My Bladder Female
Are There Any Medical Restrictions Related To The Device That I Need To Be Made Aware Of
Older models had metal in the lead which precluded MRIs. However, new technologies are now MRI-compatible. Diathermy should not be done on any patient with a sacral nerve stimulator implant. Consult with your doctor before scheduling any additional therapies or tests. He or she will discuss the need to take any precautions.
Principle Of Sacral Neuromodulation
The sacral nerves serve the lower urinary tract. To activate or inhibit urinary tract reflexes, SNM delivers mild electrical impulses to the sacral nerves. Besides the local stimulation, neuromodulation also influences the higher centres of bladder control in the brain although its mechanism is not yet clear.The procedure is done in two stages under general anaesthesia. Local anaesthesia is sometimes offered.
Recommended Reading: Medication For Overactive Bladder In The Elderly
Incontinence In Alzheimer’s Disease
People in the later stages of Alzheimers disease often have problems with urinary incontinence. This can be a result of not realizing they need to urinate, forgetting to go to the bathroom, or not being able to find the toilet. These tips may help:
- Avoid drinks like caffeinated coffee, tea, and sodas, which may increase urination. But dont limit water.
- Keep hallways clear and the bathroom clutter-free, with a light on at all times.
- Provide regular bathroom breaks.
- Use underwear that is easy to get on and off, and absorbent briefs or underwear for trips away from home.
Visit Alzheimers Disease: Common Medical Problems for more tips.
What Are The Risks Of Sns
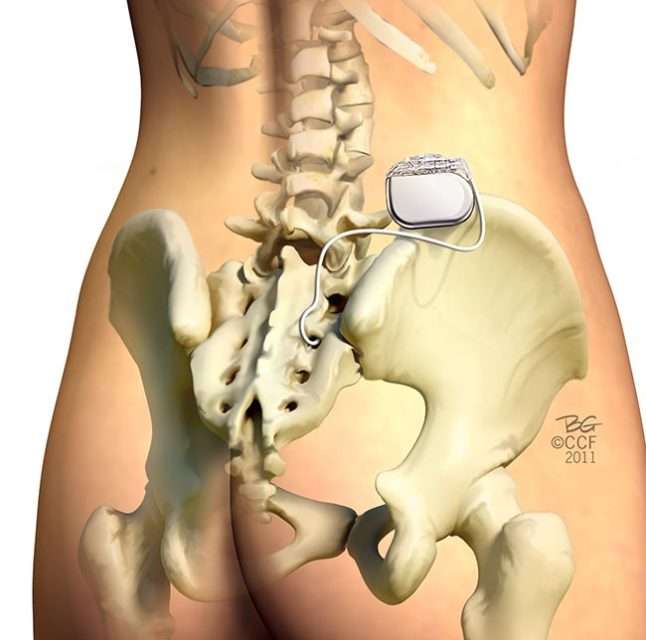
You may have pain, discomfort and infection in the area where the stimulator was implanted. You may have an allergic reaction to the materials that the lead or stimulator is made from. You may have pain or feel a shock during stimulation. The lead may move and you may need to have the procedure done again. You cannot have an MRI of your abdomen or pelvis if you have a stimulator implanted. An MRI can cause the leads to heat up and cause problems with your stimulator. You may set off metal detectors at airports or theft detectors in stores.
Read Also: Bladder Control Medication For Elderly