Fda Grants Atezolizumab Accelerated Approval As Initial Treatment For Certain Patients With Advanced Bladder Cancer
On 17 April 2017, Genentech, a member of the Roche Group, announced that the US Food and Drug Administration granted accelerated approval to atezolizumab for the treatment of patients with locally advanced or metastatic urothelial carcinoma who are not eligible for cisplatin chemotherapy. Atezolizumab was previously approved for patients with locally advanced or mUC who have disease progression during or following any platinum-containing chemotherapy, or within 12 months of receiving chemotherapy before surgery or after surgery .
Bladder cancer is the most common type of urothelial carcinoma, and up to half of all patients with the advanced form of the disease are unable to receive cisplatin chemotherapy as an initial treatment and therefore have a high unmet medical need. Urothelial carcinoma also includes cancers of the urethra, ureters and renal pelvis.
The FDAs Accelerated Approval Program allows conditional approval of a medicine that fills an unmet medical need for a serious condition, based on early evidence suggesting clinical benefit.
The indication for atezolizumab is approved under accelerated approval based on tumour response rate and duration of response. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trials. The latest approval of atezolizumab is based on the phase II IMvigor210 study.
About the IMvigor210 study
Legal
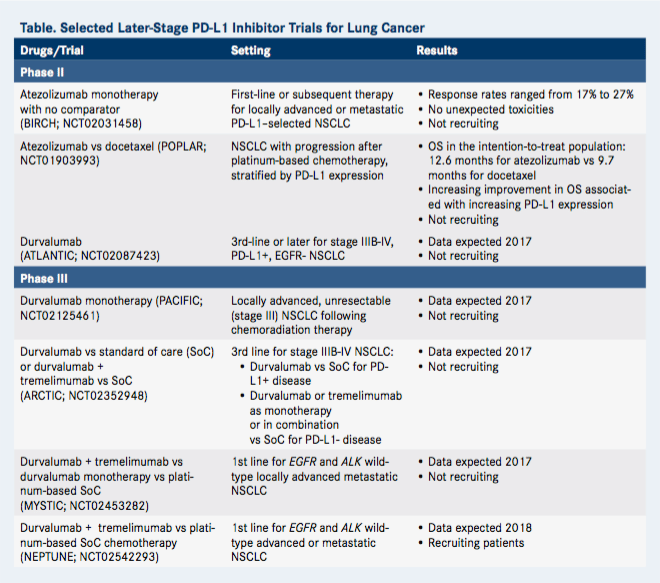
Small Cell Lung Cancer
- FDA: Recommended dosage of Tecentriq is 840 mg every 2 weeks, 1200 mg every 3 weeks, or 1680 mg every 4 weeks. Administer Tecentriq prior to chemotherapy when given on the same day until disease progression or unacceptable toxicity.
- NCCN: carboplatin AUC 5 day 1 and etoposide 100 mg/m2 days 1, 2, 3 and atezolizumab 1200 mg day 1 every 21 days for 4 cycles followed by maintenance atezolizumab 1680 mg day 1, every 28 days. Four cycles of therapy are recommended, but some individuals may receive up to 6 cycles based on response and tolerability after 4 cycles. Not recommended for relapsed disease in persons on maintenance atezolizumab at time of relapse. For persons who relapse after greater than 6 months of atezolizumab maintenance therapy, NCCN recommends re-treatment with carboplatin plus etoposide alone.
Tecentriq Receives New Indication For First
Urothelial carcinoma is the most common subtype of bladder cancer, accounting for more than 90% of bladder cancer diagnoses in the United States.1 In 2017, more than 79,000 bladder cancer cases were diagnosed in the United States, and more than 16,000 people died from this disease.2 The prognosis is favorable for patients with localized disease, with a 5-year relative survival rate of 78% for all stages of bladder cancer, which decreases to 5% for patients with distant disease.2
Lung cancer is the second most frequently diagnosed cancer in the United States, with an estimated 234,030 new cases projected for 2018.3 Nonsmall-cell lung cancer accounts for approximately 85% of all cases of lung cancer.4
Tecentriq Approved for First-Line Treatment of Urothelial Carcinoma
On April 17, 2017, the US Food and Drug Administration granted accelerated approval to atezolizumab , a monoclonal antibody intravenous inhibitor of programmed-cell death ligand-1 , as detected by the FDA-approved test, as a front-line treatment for patients with locally advanced or metastatic urothelial carcinoma who are not eligible for cisplatin chemotherapy.5
In October 2016, the FDA approved atezolizumab for the treatment of metastatic NSCLC that progressed during or after platinum-containing chemotherapy.9 Patients with NSCLC and EGFR or ALK mutations should have disease progression during FDA-approved treatment for these mutations before receiving atezolizumab.9
Mechanism of Action
Adverse Reactions
Also Check: How Can I Treat A Bladder Infection
Checkpoint Inhibitors And Bladder Cancer
Long-term survival for people diagnosed with advanced bladder cancer is poor, with approximately 5% of patients with metastatic bladder cancer surviving for 5 years or more.
Checkpoint inhibitors have shown activity in patients with metastatic bladder cancer in both the second-line setting and the first-line setting , explained Andrea B. Apolo, M.D., who heads the Bladder Cancer Section in NCIs Center for Cancer Researchs Genitourinary Malignancies Branch.
But we still needand are awaitingthe results of ongoing randomized trials comparing checkpoint inhibitors and chemotherapy in the first-line setting for patients with metastatic bladder cancer, she continued. The results will allow us to adequately compare patient outcome in terms of survival and quality of life with these therapies.
With the exception of pembrolizumab, the drugs covered by these approvals target a protein known as PD-L1 that is expressed at high levels on some cancer cells. Pembrolizumab targets PD-1, the receptor protein for PD-L1, on immune cells. Normally, binding of PD-L1 to PD-1 tamps down immune activity. By preventing the interaction between PD-L1 and PD-1, all four drugs can allow the immune system to be more active against tumor cells.
The other checkpoint inhibitor approved by the FDA for the treatment of patients with bladder cancer, nivolumab , targets PD-1.
Use In Specific Populations
Atezolizumab can cause fetal harm. Women of reproductive age should use effective contraception during atezolizumab therapy and for 5 months after the last dose. Female fertility may be compromised with atezolizumab therapy.8
Women should not breastfeed while using atezolizumab and for at least 5 months after the last dose.8
Atezolizumab has not been studied in children. No differences in its safety and effectiveness were observed between patients aged 65 years with urothelial carcinoma and younger patients, or between patients with NSCLC aged 75 years and younger patients.8
No dose adjustment is needed in patients with renal impairment or with mild hepatic impairment. Atezolizumab has not been studied in moderate or severe hepatic impairment.8
Don’t Miss: Best Thing For Bladder Infection
Fda Approves Atezolizumab For Bladder Cancer Immunotherapy
A game changing new immunotherapy drug, atezolizumab, has won FDA approval for treatment of patients with metastatic urothelial carcinoma, a type of bladder cancer.
The drug is the first new treatment for this form of the disease in more than 20 years, and is expected to have a major impact on bladder cancer, the fifth most common form of cancer and the fourth most common in men. Its benefit appears to be lasting in some patients.
Individuals diagnosed with advanced bladder cancer have not traditionally had effective treatment options. Although chemotherapy can temporarily hold the disease at bay, it often comes back within months.
Now, the US FDA has approved atezolizumab for patients with metastatic urothelial carcinoma. The drugs approval stemmed largely from results of a phase II clinical trial led by Memorial Sloan Kettering medical oncologist Jonathan Rosenberg.
Were seeing people get lasting responses with atezolizumab after chemotherapy has stopped working. Thats a game-changer for these patients, and it represents a huge breakthrough,”
says Dr. Rosenberg.
Criteria For Initial Approval
Aetna considers atezolizumab medically necessary for the following indications:
Urothelial Carcinoma – Bladder Cancer
For treatment as a single agent for bladder cancer when the requested medication is used as first line therapy in cisplatin ineligible members whose tumors express PD-L1 or in members who are not eligible for any platinum containing chemotherapy regardless of PD-L1 expression for any of the following:
Urothelial Carcinoma – Primary Carcinoma of the Urethra
For treatment as a single agent for primary carcinoma of the urethra when the requested medication is used as first line therapy for recurrent, locally advanced or metastatic disease in cisplatin ineligible members whose tumors express PD-L1 or in members who are not eligible for any platinum containing chemotherapy regardless of PD-L1 expression
Non-small Cell Lung Cancer
For treatment of NSCLC when the tumor is negative for EGFR, ALK, and RET gene mutations and any of the following criteria are met:
Read Also: Icd 10 Code For Bladder Cancer
Atezolizumabs Indication In Previously Treated Metastatic Bladder Cancer Is Withdrawn
3/31/2021 3:32:05 PM
Genentech, a member of the Roche Group, announced on March 8 that the company is voluntarily withdrawing the U.S. indication for atezolizumab in patients with prior platinum-treated metastatic urothelial carcinoma. This decision was made in consultation with the U.S. Food and Drug Administration as part of an industry-wide review of accelerated approvals with confirmatory trials that have not met their primary endpoints and have yet to gain regular approvals.
Atezolizumab is a monoclonal antibody designed to bind with the protein PD-L1, expressed on tumor cells and tumor-infiltrating immune cells, thereby blocking its interactions with both PD-1 and B7.1 receptors. By inhibiting PD-L1, atezolizumab may enable the reactivation of T cells.
The FDAs Accelerated Approval Program allows conditional approval of a medicine that fills an unmet medical need for a serious condition, with specific postmarketing requirements to confirm the clinical benefit and convert to regular approval.
Genentech will work with the FDA over the coming weeks to complete the withdrawal process. This decision does not affect other approved indications for atezolizumab.
Genentech is notifying health-care professionals about this withdrawal. Patients being treated with atezolizumab for prior platinum-treated metastatic urothelial carcinoma should discuss their care with their health-care provider.
Roche Withdraws Tecentriq For Bladder Cancer After Fda Accelerated Approval Review
This article has been updated to note that atezolizumab is approved in the US for first-line metastatic bladder cancer and that the confirmatory study, IMvigor211, did not meet its primary endpoint rather than the IMvigor 210 trial as previously reported.
NEW YORK Roche said on Monday it will withdraw the indication of atezolizumab in previously treated bladder cancer after a review of accelerated approvals by the US Food and Drug Administration.
Roche is voluntarily withdrawing the bladder cancer indication because the confirmatory study for the accelerated approval, IMvigor211, did not meet its primary endpoint in patients who were previously treated with platinum chemotherapy. As the treatment landscape for bladder cancer has advanced, atezolizumab does not fill an unmet medical need as required by the accelerated approval program, and Roche is “withdrawing this indication in recognition of the principles of the Accelerated Approval Program,” the company said.
Atezolizumab was granted accelerated approval for bladder cancer by the FDA in 2016 based on results from the IMvigor210 trial. Full approval was contingent upon positive results from the IMvigor211 study, however, that trial showed the drug did not improve overall survival in the PD-L1-high patient population.
You May Like: How To Train Bladder To Hold More Urine
Based On Fda Review Manufacturer Withdraws Atezolizumab For Use In Bladder Cancer
On March 8, Genentech, a member of the Roche Group, has announced it will voluntarily withdraw the U.S. indication for atezolizumab in patients with metastatic urothelial carcinoma who had received prior platinum therapy. The decision is based on the U.S. Food and Drug Administration review of drugs that were granted accelerated approval based on small studies whose results were later not confirmed by larger trials. Of note, this decision does not affect other approved indications for atezolizumab in nonsmall cell lung cancer, small cell lung cancer, certain types of bladder cancer, a type of triple-negative breast cancer, and liver cancer.
Continued approval for this indication for atezolizumab in metastatic urothelial cancer was contingent upon the results of IMvigor211. However, the study failed to meet its primary endpoint of overall survival in the patient population with high expression of PD-L1.
The FDA approved atezolizumab for this indication in bladder cancer in 2016. A later trial required by the FDA failed to show that this agent extended survival for this patient population. This decision by Roche follows within weeks of the voluntary withdrawal of the indication for PD-L1 inhibitor durvalumab in previously treated adults with locally advanced or metastatic bladder cancer.
Atezolizumab Indication In Us Withdrawn For Previously Treated Metastatic Urothelial Cancer
As part of an industry-wide assessment of indications based on accelerated approval, Roche in consultation with the FDA has decided to withdraw the indication for atezolizumab in patients with urothelial carcinoma following platinum-based chemotherapy.
The indication for atezolizumab in patients with urothelial carcinoma who have previously received platinum-based chemotherapy has been withdrawn in the United States, according to the drugs developer, Roche.1
The decision was made in consultation with the FDA and is part of an industry-wide assessment of indications from accelerated approval that have not met requirements of confirmation. The decision to withdraw the agent for use in this patient population does not affect indications for atezolizumab in other diseases.
The Accelerated Approval Program allows people with difficult-to-treat cancers to receive certain new therapies earlier, Levi Garraway, MD, PhD, Roches Chief Medical Officer and Head of Global Product Development, said in a press release. While the withdrawal of Tecentriq for prior-platinum treated bladder cancer is disappointing, Tecentriq continues to demonstrate benefits across multiple cancer types and therefore remains a meaningful treatment option for many patients.
Accelerated approval was based on results of the phase 2 IMvigor210 trial , which showed promising responses in patients receiving second-line atezolizumab versus historical controls.3
References:
Related Content:
Also Check: Can A Bladder Infection Clear On Its Own
Atezolizumab Approaches Fda Approval In Bladder Cancer But Biomarkers Remain Unclear
Matthew Galsky, MD, explains the challenges of utilizing molecular subtyping and PD-L1 as biomarkers, the impact a potential approval of atezolizumab could have in metastatic bladder cancer, possible combinations that could increase ORR, and next steps in understanding the IMvigor study.
Matthew Galsky, MD
The PD-L1 inhibitor atezolizumab showed promising response and durable activity in patients with metastatic urothelial carcinoma, particularly in those with increased levels of PD-L1 expression, according to results of the phase II IMvigor study recently published in The Lancet.
Based on the IMvigor data the FDA has granted atezolizumab priority review as a treatment for patients with locally advanced or metastatic urothelial carcinoma who progressed during or after platinum-based chemotherapy in the metastatic setting, or whose disease worsened within 12 months of receiving platinum-based chemotherapy before or after surgery.
Study author Matthew Galsky, MD, says the IMvigor findings represent a watershed moment for the treatment of metastatic urothelial carcinoma.
In the study, IMvigor 310 patients received atezolizumab. PD-L1 expression was determined by infiltrating immune cells in the tumor microenvironment and was defined by the percentage of PD-L1positive immune cells. Patients were identified as IC0 , IC1 , and IC2/3 .
However, the role of both PD-L1 and TCGA subtyping remains unclear in this patient population, says Galsky.
Introduction To Bladder Cancer
The treatment landscape for urothelial carcinoma has undergone a renaissance. Since 2017, eight new medications have received approval from the US Food and Drug Administration for the treatment of this disease. These new therapies include five immune checkpoint inhibitors , though one subsequently withdrew its UC indication, two antibody-drug conjugates and one tyrosine kinase inhibitor .
ICIs have improved outcomes for patients with UC with multiple agents showing efficacy in different treatment settings. Currently, there is level 1 evidence to support the use of ICIs for patients previously treated with platinum-based chemotherapy, either as switch-maintenance therapy for those with non-progressive disease or some response to platinum chemotherapy or for platinum-refractory disease as a second line or salvage therapy. Additional treatment indications include for treatment-naive patients with locally advanced, unresectable or metastatic UC who are cisplatin-ineligible with high PD-L1 expression, or for patients who are platinum-ineligible , for BCG-refractory non-muscle invasive bladder cancer with carcinoma in situ and as an adjuvant therapy after extirpative surgery. Clinical trials are ongoing further exploring additional perioperative approaches and combination therapies.
Don’t Miss: Bladder Leakage Pads For Men
Fda Panel Endorses Atezolizumab For Frontline Metastatic Urothelial Cancer
In a 10 to 1 vote, the FDAs Oncologic Drugs Advisory Committee voted to support the accelerated approval of atezolizumab for the frontline treatment of patients with cisplatin-ineligible locally advanced or metastatic urothelial carcinoma.
In a 10 to 1 vote, the FDAs Oncologic Drugs Advisory Committee voted to support the accelerated approval of atezolizumab for the frontline treatment of patients with cisplatin-ineligible locally advanced or metastatic urothelial carcinoma.
Todays positive vote reaffirms that fills a significant unmet need for people with previously untreated metastatic bladder cancer, many of whom cannot tolerate standard of care chemotherapy and need additional options, said Levi Garraway, MD, PhD, chief medical officer and head of Global Product Development at Roche, the developer of atezolizumab. Having now received positive ODAC recommendations in both bladder cancer and triple-negative breast cancer, we will continue to work with the FDA on next steps for in these indications.
We believe that the accelerated approval should be maintained while we await the final read out for the IMvigor130 trial, said Charles Fuchs, MD, senior vice president and global head of oncology and hematology drug development at Genentech, during the hearing.
In explaining his decision, Madan said, I voted yes to wait for the final results. Im optimistic that the data will continue to support the best care for our patients with bladder cancer.
Fda Grants Atezolizumab Accelerated Approval For Bladder Cancer
The US Food and Drug Administration has granted the PD-L1-targeting monoclonal antibody atezolizumab accelerated approval for first-line treatment of advanced or metastatic urothelial carcinoma regardless of PD-L1 expression status in patients who are ineligible for frontline cisplatin chemotherapy, according to a press release.1
Atezolizumab was previously FDA-approved for second-line treatment for advanced urothelial carcinomas refractory to platinum-containing chemotherapies or that had progressed within 12 months of neoadjuvant or adjuvant chemotherapy.
The FDAs Accelerated Approval Program allows conditional approval of medications addressing unmet medical needs based on early evidence of clinical benefit continued approval depends on confirmatory data verifying benefit.
This accelerated approval was based on data from the phase 2 IMvigor210 clinical trial, an open-label, multicenter, single-arm study, in which 119 patients had an objective response rate of 23.5% .
RELATED: Bladder-sparing Therapy Outcomes Similar to Cystectomy in Bladder Cancer
Serious adverse events potentially associated with atezolizumab include pneumonitis, hepatitis, colitis, endocrinopathies of the pituitary, thyroid, adrenal glands, and pancreas, neuropathy, meningitis, encephalitis, uveitis, and severe infections and infusion reactions. In the IMvigor210 trial, atezolizumab was discontinued because of treatment-related toxicities in 5 of 119 patients .
Reference
Also Check: Frequent Bladder Infections In Females