Sacral Neuromodulation Therapy Results
What would you do with more time and less discomfort? Sacral neuromodulation therapy from the Axonics Therapy device is clinically-proven to help you regain control of your bladder and bowel. Over 9 out of 10 patients are satisfied with their therapy, and nearly 9 out of 10 experienced a reduction in their urinary incontinence symptoms .
Benefits Of The Axonics System
Patients considering a third-line therapy solution for overactive bladder could benefit from the following:
- The system has a rechargeable battery that lasts approximately 20 years. Typically, patients will need to recharge the devices battery once every 4 to 6 weeks for about an hour.
- The system includes a charging belt that charges the battery, and a re-charge free remote control that communicates with the stimulator to detect when it is working, and signals the patient when the battery needs to be recharged.
- The device is MRI compatible so patients are able to undergo full-body imaging without having the device removed.
- Patients undergo an external trial of the device prior to having it permanently implanted. The trial consists of having the patient wear a small, disposable device, for a short period of time, to help them determine if the therapy provides them with notable symptom reduction.
Who Is A Candidate For Treatment With Sacral Neuromodulation
InterStim® therapy is approved for usage by Medicare in Australia in people older than 18 years of age whose symptoms have not responded to medical and conservative treatments over at least 12 months due to:
- Overactive bladder due to detrusor overactivity or
- Paradoxically also in patients with urinary retention which is not due to a blockage in the urinary tract
In Australia InterStim® therapy is also approved for use in some patients with refractory faecal incontinence which has not responded to other treatments.
Don’t Miss: Can You Have A Bladder Infection Without Symptoms
Improvements To Sacral Neuromodulation
The cardiac pacemaker was the first electrical stimulator to enjoy widespread implantation in the human body. Significant improvements were achieved in cardiac pacing when devices became responsive to real-time feedback from the heart, making therapies such as implantable cardiac defibrillation possible . This feedback, known as closed loop feedback, does not currently exist for the bladder. Some investigators have demonstrated that it is possible to distinguish between 3 levels of bladder fullness in rats by attaching cuff electrodes to nerve roots carrying primary afferent neural activity from bladder mechanoreceptors. . Others have developed implantable pressure sensors that may one day be used to provide feedback to stimulation devices . Closed loop feedback has been tested in humans with spinal cord injury, demonstrating some improvements in bladder capacity and compliance when compared with continuous stimulation . Recently this concept has also been demonstrated in dogs with similar efficacy, including an increase in bladder warning time from 10 seconds to 360 seconds. Bladder capacity improved from 70mL in the dogs receiving continuous afferent stimulation to 98mL in the dogs receiving functional electrical stimulation. The capacity went up even further to 103mL in those receiving both continuous afferent stimulation and functional electrical stimulation together .
Principle Of Sacral Neuromodulation
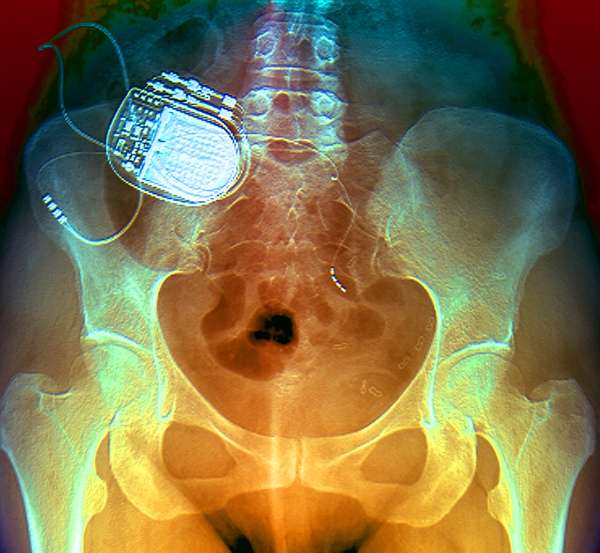
The sacral nerves serve the lower urinary tract. To activate or inhibit urinary tract reflexes, SNM delivers mild electrical impulses to the sacral nerves. Besides the local stimulation, neuromodulation also influences the higher centres of bladder control in the brain although its mechanism is not yet clear.The procedure is done in two stages under general anaesthesia. Local anaesthesia is sometimes offered.
You May Like: How To Fix Bladder Leakage Naturally
Case Snapshot: Sacral Nerve Stimulation For Medically Refractory Urinary Retention And Overactive Bladder In A Pediatric Patient
Section of UrologyCo-Director, Surgical Neuromodulation Center
Initially used as a treatment for refractory adult urinary incontinence only, neuromodulation of the sacral nerve is now approved in adults by the Food and Drug Administration as an acceptable option after other modalities have proven ineffective for the treatment of urinary and/or fecal incontinence. However, the application of this approach in pediatrics is a relatively new concept. In 2012, the Motility Center in the Division of Gastroenterology, Hepatology and Nutrition and the Section of Urology at Nationwide Childrens Hospital began offering sacral nerve stimulation to children with intractable urinary or fecal incontinence. Since then, more than 25 patients have received the implants. Here, we profile a patient whose experience illustrates the benefits of the approach adopted at Nationwide Childrens.
Who Will Benefit From Sacral Neuromodulation
Patients with chronic urinary retention, voiding dysfunction, bowel dysfunction and chronic pain syndromes can benefit from this device. Indeed, it may be the only therapy available in some patient groups. In patients with chronic urinary retention, the only known forms of treatment are clean intermittent self-catheterization or indwelling suprapubic/transurethral catheters. At times CISC can be a painful and traumatic process, and SNM offers an effective therapeutic alternative.
Don’t Miss: What Are The Best Pads For Bladder Leakage
Pros And Cons Of Neuromodulation Therapy For Overactive Bladder
Sacral nerve stimulation is a highly effective treatment. Most people note success with the treatment. However, the devices may stop working. Approximately 2/3 of people will note that the device stops working within five years if this occurs, another surgery is required to replace the device or the battery.
It is also a process to be approved for the device. The provider will need to verify that the device will work before surgically implanting it. In order to do this, a test is performed. The test involves making a small cut in the lower back and placing a wire in this cut. The wire is attached to a battery powered stimulator, which is carried for three weeks. If the trial is successful, then permanent placement can occur.
It is also important to remember that placement of the stimulator requires a surgery. Having any surgery carries risks, such as infection and pain. Other side effects of the placement of SNS include:
- Temporary electric shock feeling
- Bleeding at the implant site
Other stimulation options are available. For example, percutaneous tibial nerve stimulation involves insertion of a thin needle near the tibial nerve. A stimulator sends impulses through this needle to the nerve, which sends the impulses to the bladder. PTNS treatments are approximately 30 minutes long and require about 12 treatments, though more sessions may be required. There are minimal side effects, but they include:
- Tingling or mild pain
- Bruising or bleeding at the insertion site
What Does Treatment With Sacral Neuromodulation Involve
The InterStim® device works like a pacemaker for the bladder in a similar way that a cardiac pacemaker works for the heart.
Surgery to insert the InterStim® device involves a two staged procedure.
Stage 1 the trial stage involves the insertion of a pacing wire alongside a sacral nerve using X-ray guidance as a day case procedure.
- If the trial period is successful in reducing urinary symptoms of bladder overactivity by at least 50%, the patient can proceed to implantation of the full pacemaker device.
- The advantage of the trial stage is the ability to test out the InterStim® device over several days to see if it suits the individual before committing to the treatment.
- About 7 in 10 patients choose to move from the test stimulation to have the permanent InterStim® pacemaker inserted.
Images supplied courtesy of Medtronic Inc.
Stage 2 the insertion of the permanent InterStim® pacemaker involves the implantation of a device smaller than the size of a box of matches into the fat of the buttock where it cannot be seen.
Images supplied courtesy of Medtronic Inc.
Read Also: Can Clindamycin Treat Bladder Infection
What Is Sacral Neuromodulation
Neuromodulation for Overactive Bladder is a therapy in which small electrical signals stimulate the nerves associated with urine storage. These signals change the way that the bladder sends messages to the brain. In OAB, it is those signals which are overactive and neuromodulation can help to decrease them. Percutaneous Tibial Neuromodulation is one way of stimulating those nerves. However, right now patients have to come into the office for every PTNM treatment. This is difficult for many patients. Sacral neuromodulation also stimulates the bladder nerves to decrease feelings of bladder urgency, but can be implanted so that a patient is treated continuously without such frequent visits.
Sacral neuromodulation stimulates the nerve that controls the bladder by placing a tiny wire through a small, natural, opening in the sacrum. The sacrum is a the large, shield-shaped bone in your pelvis . The wire is placed in this location for several reasons. The sacrum provides safe access to the bladder nerves because the nerve is relatively close to the skin in this location and there are no body structures between the skin and the nerve that could be injured by the wire. The sacrum also provides a stable attachment point so that the lead is held firmly in place. Finally, the sacrum is close to the buttocks which allows the neuromodulation battery to be buried deep in the fat.
Patient: Medically Refractory Urinary Issues
Phase I: Case HistoryDue to vesicoureteral reflux and recurrent UTIs, a 3-year-old female underwent ureteral reimplantation surgery in 2004. She was later managed and treated by gastroenterologists and urologists at Nationwide Childrens for overactive bladder symptoms and chronic constipation. Multiple anticholinergic medications failed to resolve the overactive bladder symptoms, and laxatives and bowel stimulants did not effectively treat the constipation. Due to poor bladder emptying, she had performed twice daily self-catheterization to assist with post-void residuals since July 2011. Persistent symptoms interfered with schoolwork and sports participation.Phase II: Presentation and TreatmentAt 12 years of age, the child presented to the author in June 2013 for ongoing urinary incontinence, overactive bladder symptoms, poor bladder emptying, UTIs and constipation. Previous MRI of the spine and anorectal manometry were normal. Her most recent urodynamics study showed multiple uninhibited bladder contractions during filling, indicating an overactive bladder as well as a small functional bladder capacity. She voided with an intermittent straining pattern and had a large post-void residual volume. She underwent placement of a temporary sacral nerve stimulator in July 2013. A permanent device was implanted two weeks later due to a marked improvement in symptoms.
You May Like: Best Over The Counter Bladder Infection Medicine
How Does Sacral Nerve Stimulation Actually Work
Sacral nerve stimulation therapy uses a small device that is implanted under the skin in the upper buttock area. The device sends mild electrical impulses through a lead that is positioned close to a nervelocated in the lower back , which influences the bladder,the sphincter and the pelvic floor muscles.
A neurotransmitter device, implanted under the skin in the upper buttock area, transmits mild electrical impulses through a lead wire close to the sacral nerve. The impulses, in turn, influence the bladder sphincter and pelvic floor muscles providing bladder control.
Watch Dr Elizabeth Mobleys Webinar Titled Its Time To Regain Control Of Your Bladder And Bowel This Video Was Hosted And Produced By Axonics Inc
Axonic Modulation Technologies has shared the following educational resource materials for patient review. Please note: Some of the materials supplied by Axonics references bowel incontinence. Urology Austin providers do not treat bowel incontinence. If youre struggling with overactive bladder or urinary retention, contact Urology Austin to schedule an appointment with one of our providers to see if the Axonics System is right for you.
You May Like: What Kind Of Doctor Treats Bladder Problems
Quality Assessment Of The Included Literature
Level of evidence was stated per article as defined in publications of the University of Oxfords Centre for Evidence-Based Medicine . Risk of bias assessment was performed according to the Cochrane Collaboration guidelines .
Table 1.
Level of evidence and risk of bias
Most articles described cohort study designs . The remainder were case series and randomized controlled trials . In most studies, primary outcome was a successful SNM test result defined as a 50% improvement in urinary symptoms. In 34 studies, test stimulation success was the main outcome . In 19 studies, both test stimulation outcome and permanent SNM outcome were investigated . In 14 studies, only permanent SNM outcome was investigated .
In 15 studies, a percutaneous nerve evaluation was performed and in 23, a tined lead procedure . In 19 studies, patients underwent either PNE or TLP. In 3 studies, it was unknown which procedure was used . In most articles, risk of bias domains was assessed as low risk or unclear risk . A graphical summary of the results is depicted in Figure 3.
Table 2.
Risk of bias assessment .
Fig. 3.
a-g Graphical summary of the result. SNM, sacral neuromodulation PNE, percutaneous nerve evaluation TLP, tined lead procedure UUI, urge urinary incontinence.
Axonics Announces First Patient Implants In Canada With New Recharge
IRVINE, Calif.—- Axonics, Inc. , a global medical technology company that is developing and commercializing novel products for the treatment of bladder and bowel dysfunction, today announced the first patient implants in Canada with the Axonics F15, the companys newly developed, long-lived, fully recharge-free sacral neuromodulation system.
The University of Alberta implanted four patients this week with the Axonics F15. The four patients were programmed at stimulation amplitudes between 0.95mA and 0.30mA, resulting in an expected battery longevity in the body ranging from 18 years to over 22 years.
The Axonics F15 received regulatory approval from Health Canada following U.S. FDA approval. The comprehensive U.S. commercial launch of the Axonics F15 commenced in the second quarter of 2022.
The approval of the Axonics recharge-free system is welcome news for patients in Canada suffering from bladder and bowel dysfunction, said Gary J. Gray, M.D., FRCS, a urologist and clinical professor at the Kipnes Urology Centre in the Department of Surgery at the University of Alberta. I am excited to now offer patients a recharge-free option that significantly increases device longevity in the body alongside the rechargeable Axonics SNM system that has helped so many of my patients.
About the Axonics F15 SNM system
About Axonics
View source version on businesswire.com:
Contacts
Recommended Reading: Back Pain Causing Bladder Problems
Sacral Neuromodulation And Interstim For Overactive Bladder
Sacral neuromodulation is regarded as a 3rd line treatment for patients with overactive bladder i.e. after medications and bladder retraining with pelvic floor physiotherapy have been unsuccessful in controlling symptoms.
Patients are regarding as having refractory symptoms when they have not responded to or do not tolerate at least 2 of the newer medications available for OAB symptoms.
How Successful Is Sacral Neuromodulation
One way of looking at the success of SNM is how many patients have a successful test and have the device permanently implanted. Approximately 70-85% of patients see this much improvement. However, it is important to remember that OAB is a chronic disease and you likely want to know how well the treatment performs over time. Like all OAB therapies, it appears there is some drop in success rates over time. One study demonstrated a decline to 62% success rate at 5 years. Still, for a majority of patients, SNM remains a successful and satisfying therapy for many years. In studies comparing older and younger patients, there does not appear to be any significant difference in how well older patients respond to therapy.
Don’t Miss: Bladder Cancer In Cats Treatment
Living With An Overactive Bladder
An OAB can affect all aspects of your life. Its not only physically uncomfortable, but it involves making many trips to the bathroom, which some may find embarrassing. People with an OAB may experience relationship issues, difficulty sleeping, issues with their sex lives, and other mental health challenges related to their daily experiences.
But youre not alone, and help is out there. Along with treatments aimed at lessening your symptoms, resources are available to people living with an OAB. Some organizations that can help you find help and support include:
Is Neuromodulation Therapy An Effective Way To Treat Overactive Bladder
Rather than a specific condition, overactive bladder is a collection of symptoms used to describe difficult to manage urinary issues and bladder problems, such as a frequent and sudden urge to urinate. It can be difficult to control. Urinary incontinence may also be experienced, which causes an unintentional loss of urine.
Recommended Reading: Nerve Stimulation For Overactive Bladder
What Are The Risks Of Sacral Neuromodulation
The most significant risk associated with sacral neuromodulation is infection of the device. An infected sacral neuromodulation device must be removed entirely. A new device can be place after a suitable time to clear the infection and heal but this is a very expensive and distressing complication. Historically, rates of removal for infection have ranged from 3-10% but more recent evidence suggests infection rates can be around 1% or even lower. Several studies have looked at increased rates of infection in certain types of patients, such as those with diabetes, in an effort to decrease infections. In my own practice, over the last few years, I developed an infection prevention program which appears to reduce the rate of device infections by nearly 90%. Infections, however, have not been entirely eliminated even with this protocol.
What Are The Side Effects
The SNM procedure is very safe and has not been associated with any death. The majority of adverse events experienced were of a relatively minor nature. In a report to the Medical Services Advisory Committee of Australia:
-
The most commonly reported clinical adverse events were
-
Pain of undefined location and severity
-
Pain specifically at the implanted pulse generator site
-
Lead migration was reported at 7%
-
Approximately 16 % of patients required lead revision or replacement, frequently in order to optimise the clinical effectiveness of the device
-
Device removal was reported at a rate of 9.9% in the implanted patients. This is usually done because of infection, lost efficacy, or pain.
If you want to know whether sacral neuromodulation can help your bladder problems, contact us.
For more specific information on the Testing Phase procedures and instructions and the Implant Phase read Sacral Neuromodulation – User Info.
Read Also: Does Coffee Cause Bladder Infections